Research
Our group is currently focusing on two main research topics:
- Design and synthesis of glycomimetics
- Allosteric modulators of the Hsp90 chaperone protein
Glycomimetics
The interaction of glycans with sugar-binding proteins (or lectins, see here) is at the heart of a large number of pathological and physiological events. Our goal is to gain control of these processes by means of molecules that can interfere with the sugar-protein interaction step.
Many structures have been suggested to mimic monosaccharides (mostly in the context of glycosidase inhibition). Less is known about mimicking oligosaccharides with antagonists able to block their interaction with sugar-binding proteins.
Our approach is based on the observation that many bioactive oligosaccharides can be dissected into one relatively small pharmacophoric portion, which actually interacts with the biological target, and one large seemingly inactive scaffold formed by the oligosaccharide backbone, which appears to be holding the pharmacophore in place for appropriate interaction.
We seek to simplify these structures by replacing their complex scaffolds with simpler molecules designed to maintain the required orientation of the pharmacophoric portion and to improve the metabolic stability of the resulting construct. In this way we hope to build so-called “drug-like” properties in our glycomimetics, and make them useful as potential drugs.
We are currently involved in the European Training Network IMMUNOSHAPE, a project that aims to developing selective immunomodulators targeting C-type Lectin Receptors (CLR) on Antigen Presenting Cells. In this context, we are generating a library of printable glycomimetic compounds, which will be screened in an array format against relevant C-lectins.
DC-SIGN antagonists as inhibitors of viral infections
DC-SIGN is a tetrameric lectin receptor of dendritic cells, involved in sexual transmission of HIV. Blocking DC-SIGN may allow to block viral entry at the mucosal level and represents a new opportunity for developing more effective antiviral drugs.
In a collaborative effort with the European Research Training Network Carmusys, we showed that the tetravalent presentation of the linear pseudo–trimannoside 1 on a polyester scaffold yields an active dendron (3) which can inhibit the trans infection of CD4+ T-lymphocytes by HIV mediated buy DC-SIGN. This dendron presents negligible cytotoxicity and a long-lasting effect both in cellular models and on cervical tissue explants. (ACSChemBiol 2010, 301-312; AIDS 2012, 26, 127-137)
Higher valency presentations on similar polyester dendrimers led to very potent inhibitors (in the nanomolar range) of cell infection by Ebola pseudotyped viral particles (Bioconj. Chem. 2011, 22, 1354–1365). Surprisingly, equivalent constructs obtained from pseudo-di- (2) or pseudo-tri- (1) saccharides displayed similar potency, even if, in SPR competition experiments, 1 was found to be an order of magnitude more active than 2. The combination of several biophysical techniques revealed that the reason for this puzzling observation is the ability of 1 to cluster DC-SIGN tetramers in solution, which leads to an overestimation of its inhibitory potency in the SPR assay (ACS Chem. Biol. 2014, 9, 1377).
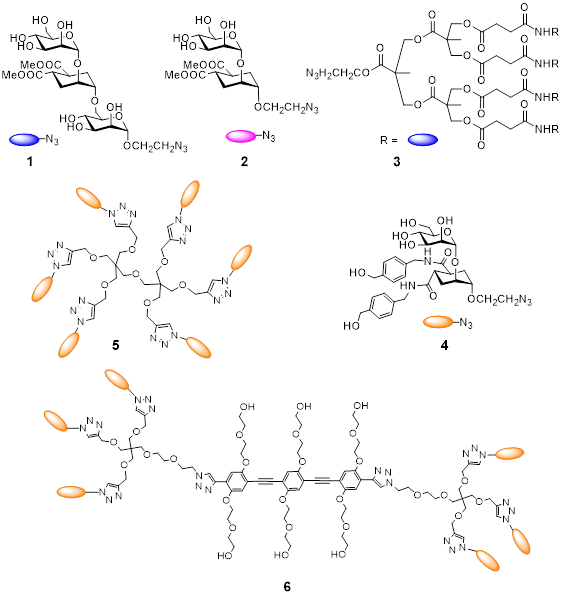
Monovalent and polyvalent glycomimetic compounds designed and synthesised in our group:
Pseudo-triMan 1, Pseudo-diMan 2, tetravalent dendron 3 ( a.k.a. Polyman2),
hexavalent constructs 5 (Polyman 19, PM19) and 6 bear the optimized ligand 4 (Man30).
Optimization of the monovalent ligand (4, Chem. Eur. J. 2013, 19, 4786) and of the polyvalent scaffold led initially to dendrimer 5 (Biomaterials 2014, 35, 4175) and finally to the elongated version 6 (Chem.Commun. 2015, 51, 3816), which inhibits DC-SIGN mediated HIV infection with an IC50 of 24 nM. The high potency of the latter compound depends on its ability to span two adjacent binding sites in the DC-SIGN tetramer.
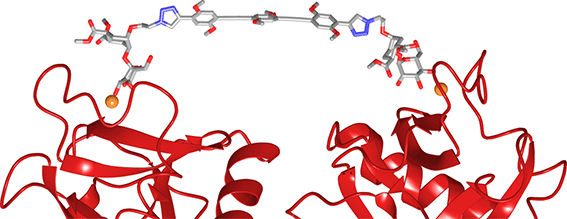
Divalent version of 6 (Polyman X) spanning two adjiacent binding sites in the DC-SIGN tetramer
Potential antiviral compounds targeted against DC-SIGN were synthesized also using a common fucosylamide anchor (Org. Biomol. Chem. 2011, 9, 5778-7705; Eur. J. Org. Chem. 2013, 5303). A potent DC-SIGN targeting device was developed using gold nanoparticles functionalized with α-fucosyl-β-alanyl amide (Bioconj. Chem. 2014, 25, 2093).
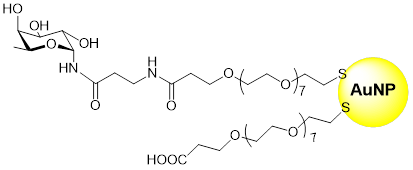
Fucosylated gold nanoparticles (GNPs)
Antagonists of the Mannose Binding Lectin (MBL)
In this project we are collaborating with neurobiologists at the Mario Negri Institute for Pharmacological Research to investigate the pivotal role of mannose-binding lectin (MBL), one of the recognition molecules of the lectin complement pathway, in brain ischemic injury and trauma.Our data show an important role for MBL in the pathogenesis of brain ischemic injury and provide a strong support to the concept that MBL inhibition may be a relevant therapeutic target in humans, one with a wide therapeutic window of application. (Circulation 2012, 126, 1484) Similarly, we were able to show that inhibition of MBL with appropriate mannose-based dendrimers alleviates neurobehavioral dysfunction following experimental traumatic brain injury (J. Cereb Blood Flow Metab 2016).
These results were obtained with pseudo-mannodendrimers of various structures, the latest of which was described in Chemistry Eur. J. 2016, 22, 3686
Cholera toxin inhibitors
We have developed a number of glycomimetics and neo-glycoconjugate structures that bind to the Cholera Toxin inhibiting its activity (Chemistry Eur. J. 2008, 14, 7434). Most recently, we have reported on the synthesis of a library of neo-glycoconjugates as non-hydrolizable inhibitors of the cholera toxin (Chemistry Eur. J. 2010, 1951). These molecules feature a galactose and a sialic acid as pharmacophoric residues and were synthesized from readily available precursors avoiding O-glycosidic bonds. Affinity could be enhanced up to one or two orders of magnitude over the affinity of the individual pharmacophoric sugar residues (see: pseudo-GM1 project).
