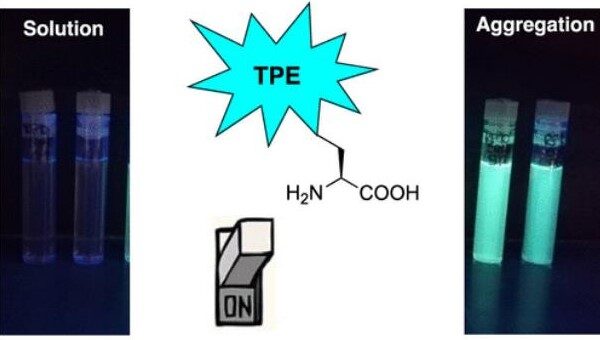
Aggregation-Induced Enhanced Emission of Tetraphenylethene-phenylalanine Hybrids: Synthesis and Characterization
Aggregation-induced emitting (AIE) luminophores are sensitive and easy-to-handle types of robes that allow driving a stimulus-responsive off/on optical tool through the manipulation of the aggregation behavior. In this work, tetraphenylethene (TPE)-phenylalanine derivatives, haracterized by...
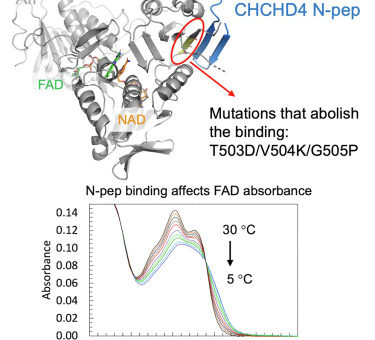
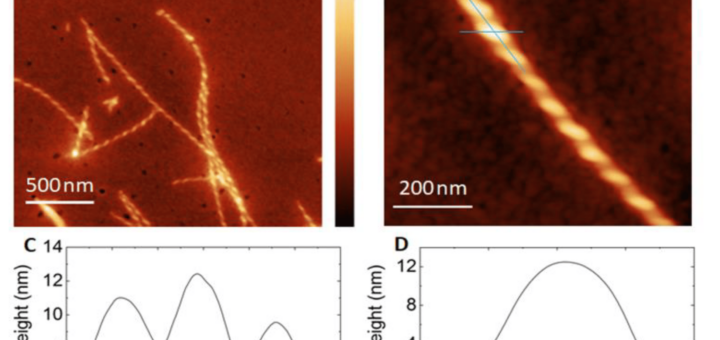
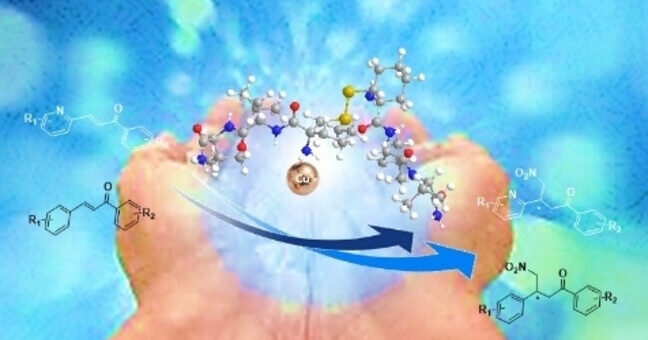
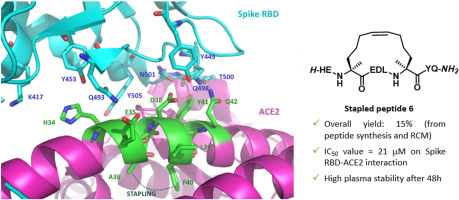






comments